Absolute vs gauge pressure instruments
- Absolute pressure is referenced to perfect vacuum. It indicates how much higher your process pressure is than that of a process in vacuum, so measurements are always positive. Absolute pressure readings are commonly used in vacuum processes, such as vacuum deposition.
- Gauge pressure is referenced to local atmospheric pressure. It indicates how much above or below local atmospheric pressure your process pressure is, so measurements can be either positive or negative.
When to use absolute pressure
In some situations, it is fairly straightforward whether absolute or gauge pressure should be used. Absolute pressure should be used in processes requiring a specific amount of pressure, regardless of what happens in the atmosphere. Applications that require measurement or control of low atmospheric pressures almost always benefit most from absolute pressure control. A couple example include back pressure control for process analyzers and flow characterization for cigarette filters.When to use gauge pressure
Gauge pressure controllers add or remove air as the ambient air pressure fluctuates, to maintain the desired pressure differential. This means gauge pressure should be used when your process requires a certain pressure relative to atmospheric pressure.
Gauge pressure is typically used in processes where pressure cannot dip below atmospheric pressure, such as when measuring and controlling tire pressure.
When either gauge or absolute pressure are viable options
Sometimes it is a little more challenging to determine which is right to use. The following examples illustrate situations where either gauge or absolute pressure could work, depending on what types of measurement and control you are looking for.
Example 1:
Changing altitude
As altitude increases, the amount of gas molecules in the air decreases leading to a decrease in atmospheric pressure. This means at different altitudes, measures of absolute pressure will give very different readings than measures of gauge pressure.
Process conditions: If you tighten the cap on an empty water bottle in Tucson (elevation: 2,160 feet, ambient air pressure: 13.67 PSIA), the pressure within the bottle will be 13.67 PSIA and 0 PSIG. If you then drive the bottle up to Mt. Lemmon (elevation: 9,159 feet, ambient air pressure: 10.44 PSIA), the absolute pressure within the bottle will be 13.67 PSIA and the gauge pressure will be 3.23 PSIG.
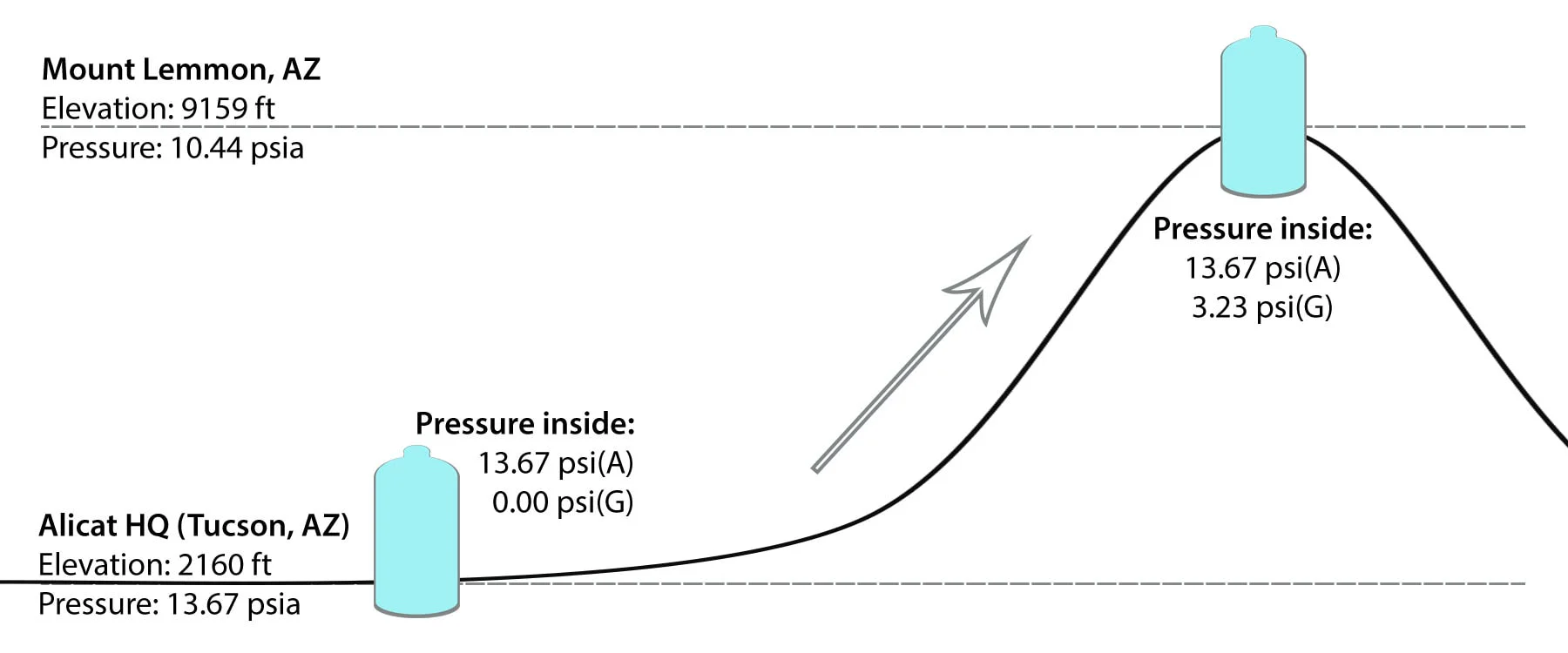
Figure 1. Gauge pressure of a process increases as atmospheric pressure decreases.
Now, imagine you open the bottle at Mt. Lemmon, and the pressure built up within the bottle releases into the air until it reaches an internal pressure of 10.44 PSIA and 0 PSIG. You then reseal the bottle, and head back to Tucson. The pressure inside the bottle is still 10.44 PSIA, but gauge pressure is now -3.23 PSIG.
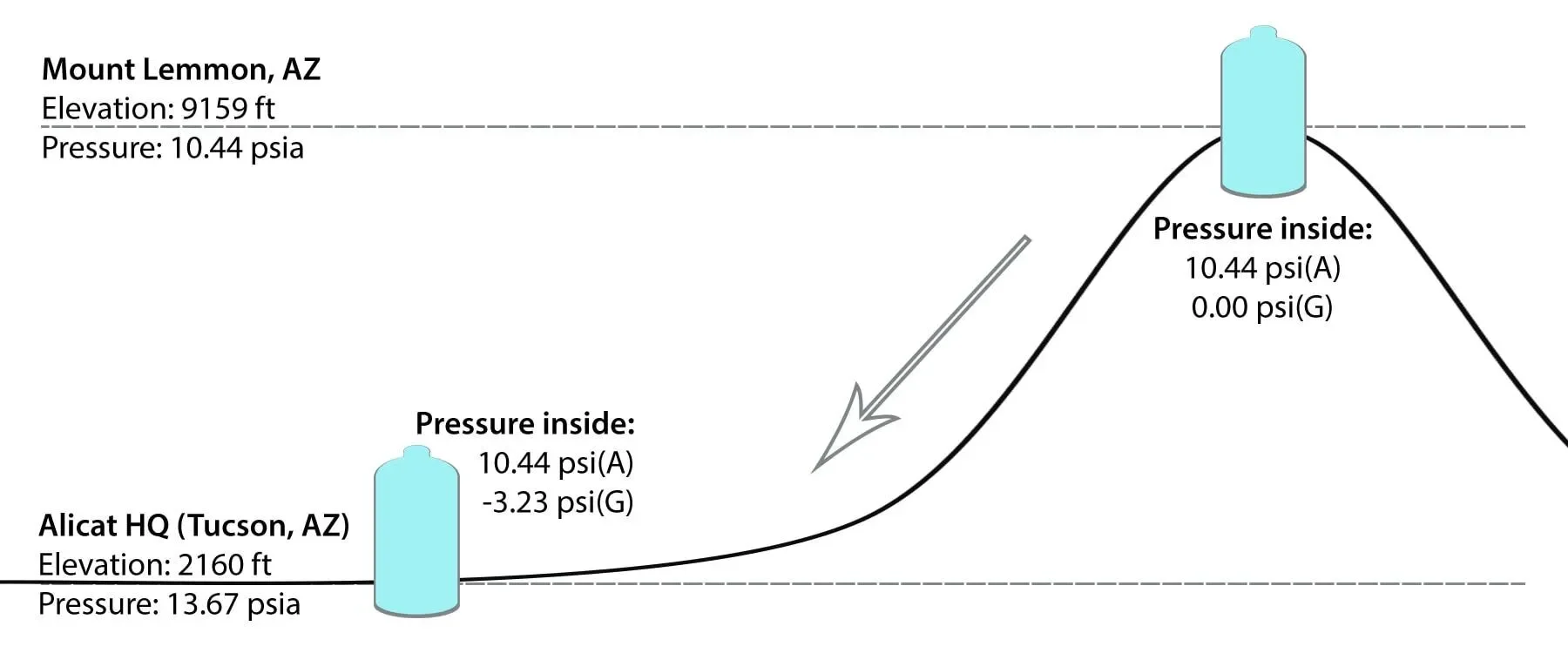
Figure 2. Gauge pressure of a process increases as atmospheric pressure increases.
Solution: Either measurement (absolute or gauge) may be useful in this case, depending on what you are looking for. It is simply important to understand what each measurement is telling you about the process, and to determine which provides the most valuable information.
Example 2:
Fluctuations in local atmospheric pressure fluctuations
Throughout the year, it is common for a single location’s atmospheric pressure to vary as much as 0.3 PSI (or more for locations with frequent storms, tropical depressions, or hurricanes).
Process conditions: In Tucson, the average atmospheric pressure is about 13.7 PSIA ± 0.1 PSIA. If a process requires being held at 0.3 PSI above atmospheric pressure, would a gauge or absolute pressure controller be better?
Solution: Gauge pressure readings would fluctuate with local atmospheric pressure variations, as in figure 3. These fluctuations would not be visible, however, because the controller would always read a gauge pressure of 0.3 PSIG. Absolute pressure readings, on the other hand, would remain steady regardless of pressure fluctuations.

Figure 1. Gauge pressure of a process increases as atmospheric pressure decreases.
It may seem like absolute pressure is the clear answer, but either could work depending on the scale. For example, fluctuations from 113.6 PSIG to 113.8 PSIG aren’t as significant as fluctuations from 13.6 PSIG to 13.8 PSIG. It really depends on what you need to measure or control, and what your error tolerances are.